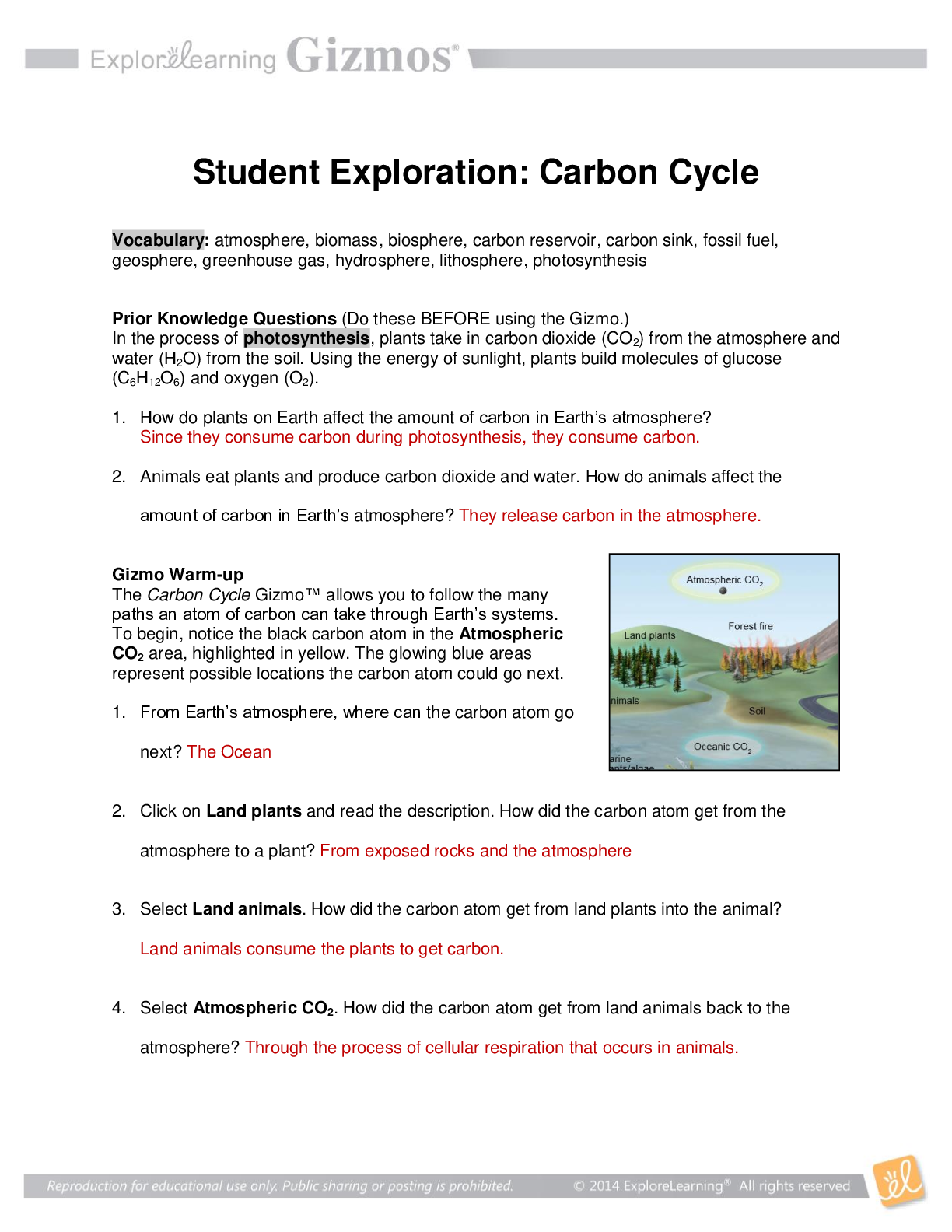
Latest Review Test 7 Submission Psych 255
$ 5

ATI MED-SURG PROCTORED LATEST FINAL EXAM 2025-2026 ACTUAL QUESTIONS WITH 100% CERTIFIED, ELABORATED & VERIFIED SOLUTIONS TOP SCORE✓✓ ✓ACE YOUR EXAMS
$ 18.5
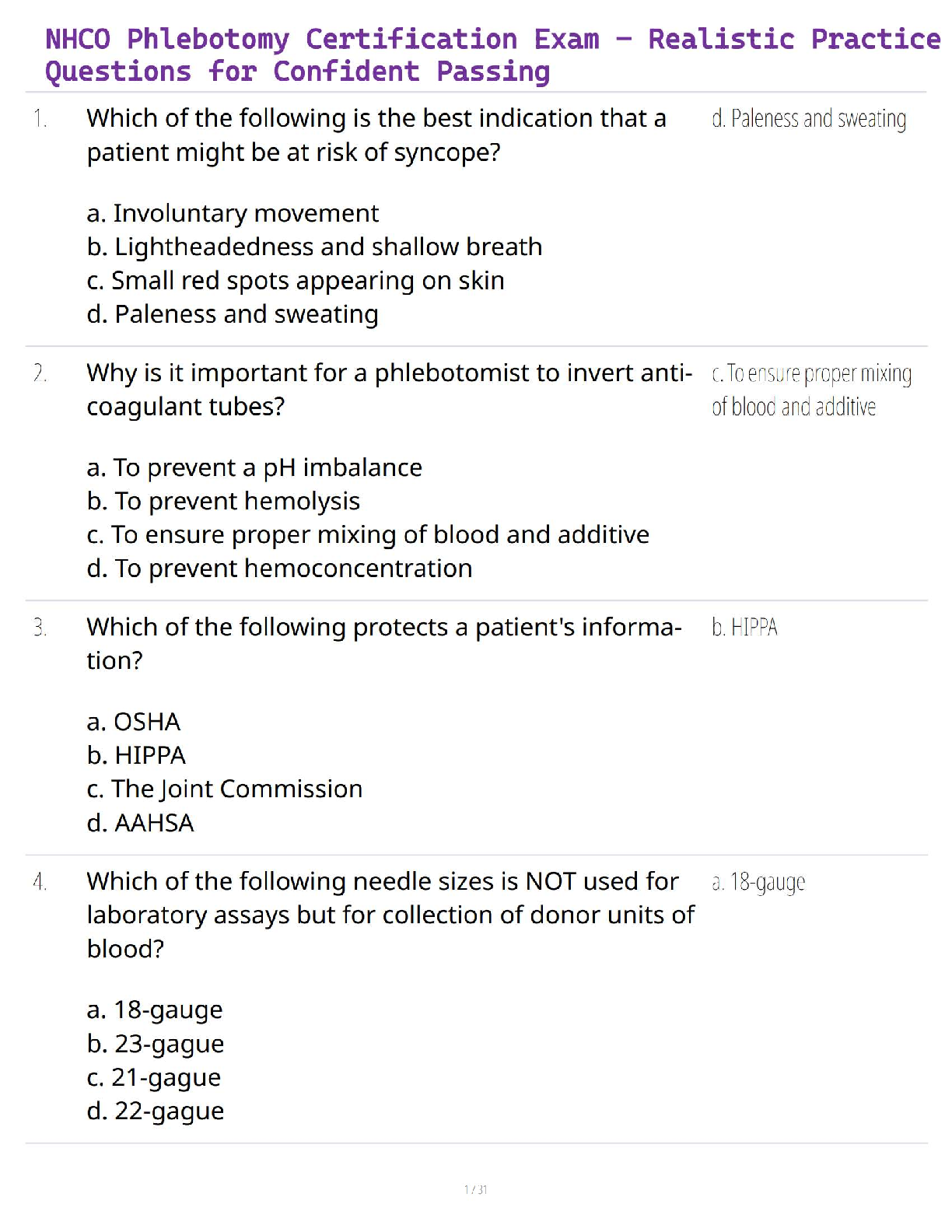
NHCO Phlebotomy Certification Exam – Realistic Practice Questions for Confident Passing
$ 21

COMPLETE INTRODUCTION OF CELLS.
$ 19
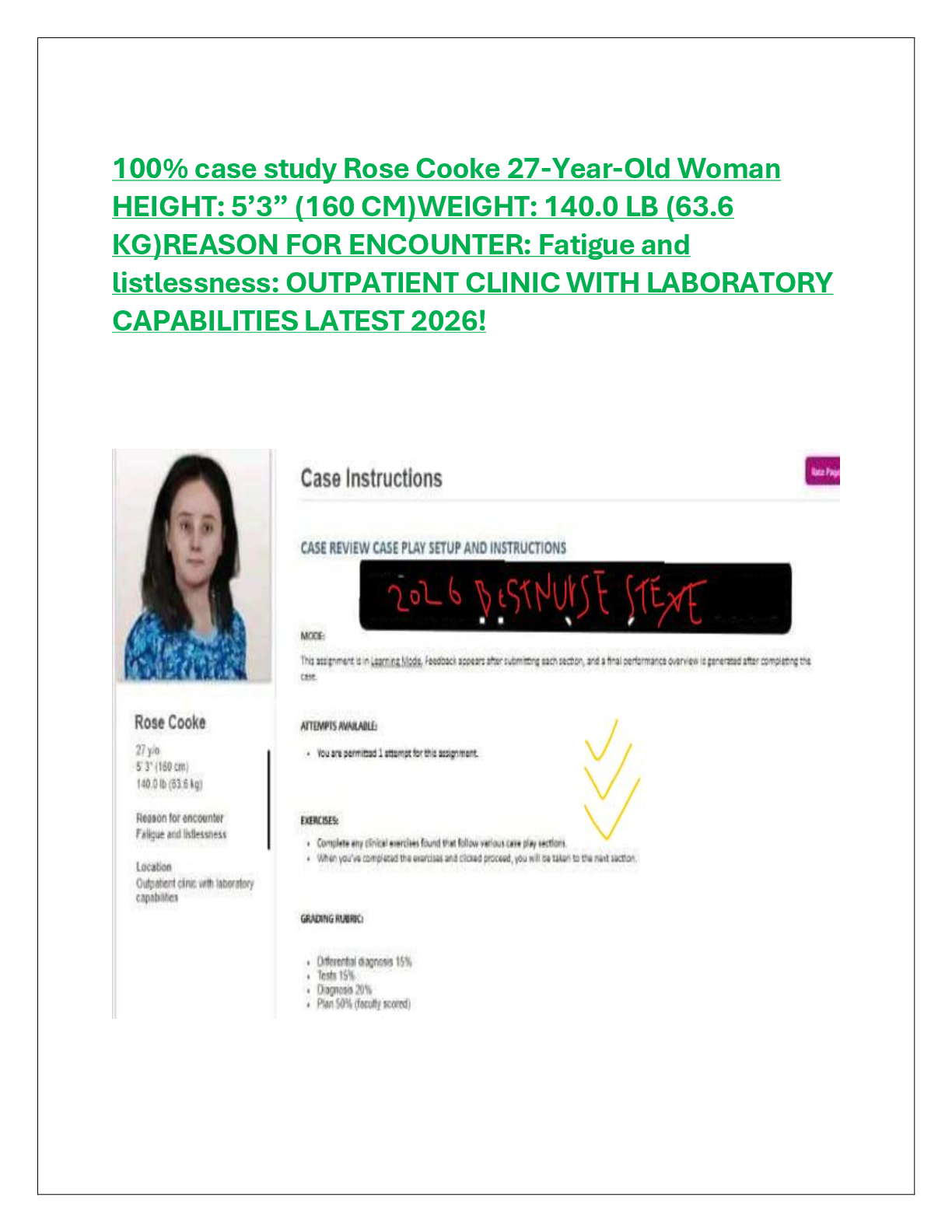
100% case study Rose Cooke 27-Year-Old Woman HEIGHT: 5’3” (160 CM)WEIGHT: 140.0 LB (63.6 KG)REASON FOR ENCOUNTER: Fatigue and listlessness: OUTPATIENT CLINIC WITH LABORATORY CAPABILITIES LATEST 2026!
$ 17

LMT1 Task 4 Measurement, Analysis, and Knowledge Management for Educational Leaders LMT
$ 5

eBook PDF Auto Suspension and Steering 5th Edition By Chris Johanson, Martin Stockel
$ 25

eBook [PDF] Qualitative Research Methods for the Social Sciences 9th Global Edition By Howard Lune, Bruce Berg
$ 25

NCLEX EXAM REVIEW QUESTIONS AND ANSWERS NEW LATEST UPDATED GUIDE 2022
$ 14

Maternal-Newborn-Nursing-Care-Best-Evidence-Based-Practices-Nursing-Workbook
$ 20

Social Sciences : Social Work (MSW)
$ 29
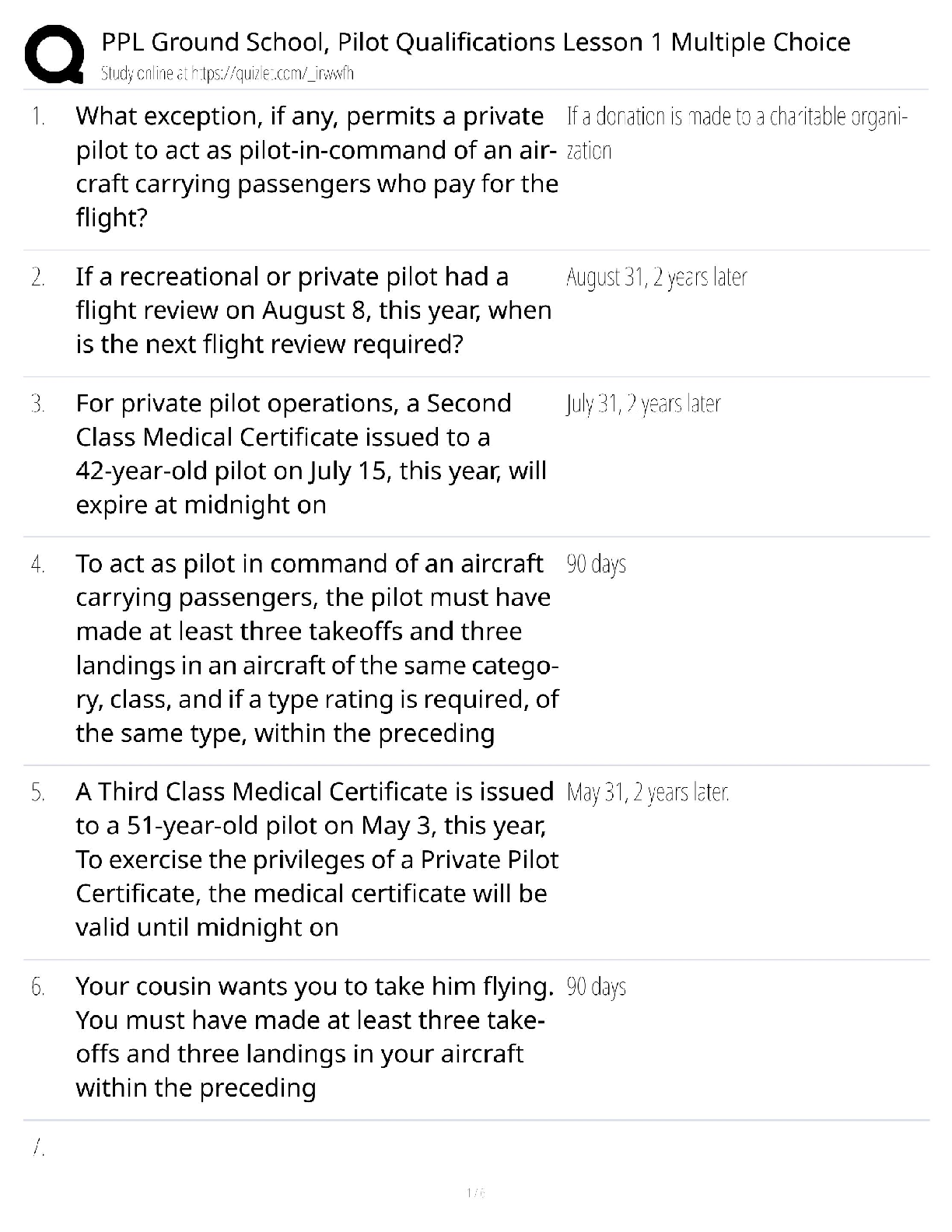
PPL Ground School Pilot Qualifications Lesson 1 Multiple Choice / Score 100% / 2025 New Version
$ 22
Reproductive-First-Aid-For-The-Usmle-Step-1-2020-Thirtieth-Edition
$ 18

Case Solutions for EMIRATES AIRLINE CONNECTING THE UNCONNECTED by Juan Alcacer John Clayton
$ 45

Pharmaceuticals The Ethics of Drug Pricing.pdf
$ 7

(C919 NURS 6002) Facilitation of Context - Based Student centered Learning - Comprehensive FA Guide Q & S 2024
$ 12

Actual 2024 AQA A-level ENGLISH LITERATURE B 7717/1B Paper 1B Question Paper + Mark Scheme Actual 2024 AQA A-level ENGLISH LITERATURE B 7717/1B Paper 1B Literary genres: Aspects of comedy Merged Question Paper + Mark Scheme
$ 7
2301.7 Defense Acquisition System / Adaptive Acquisition Framework / Score 100% (Study Guide)







.png)