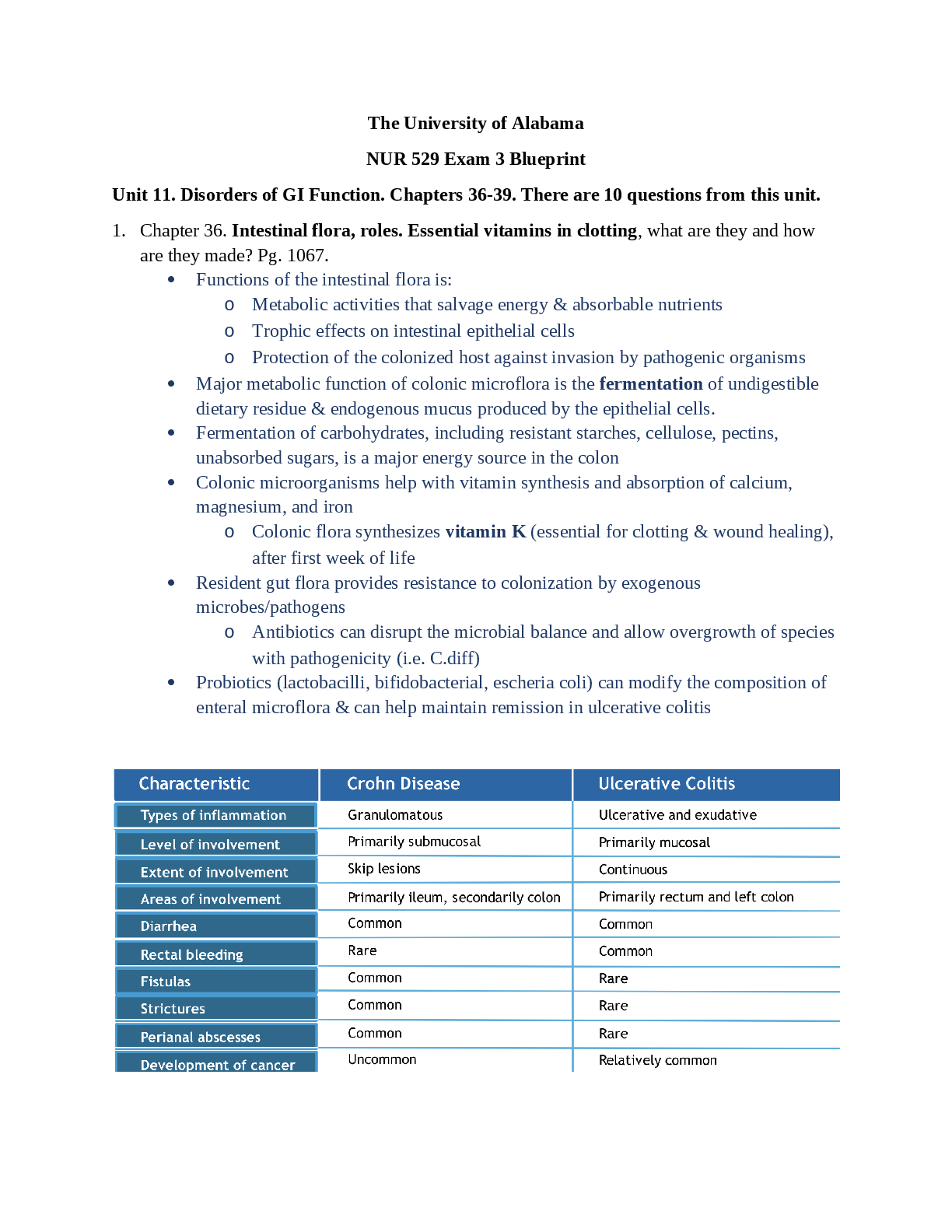
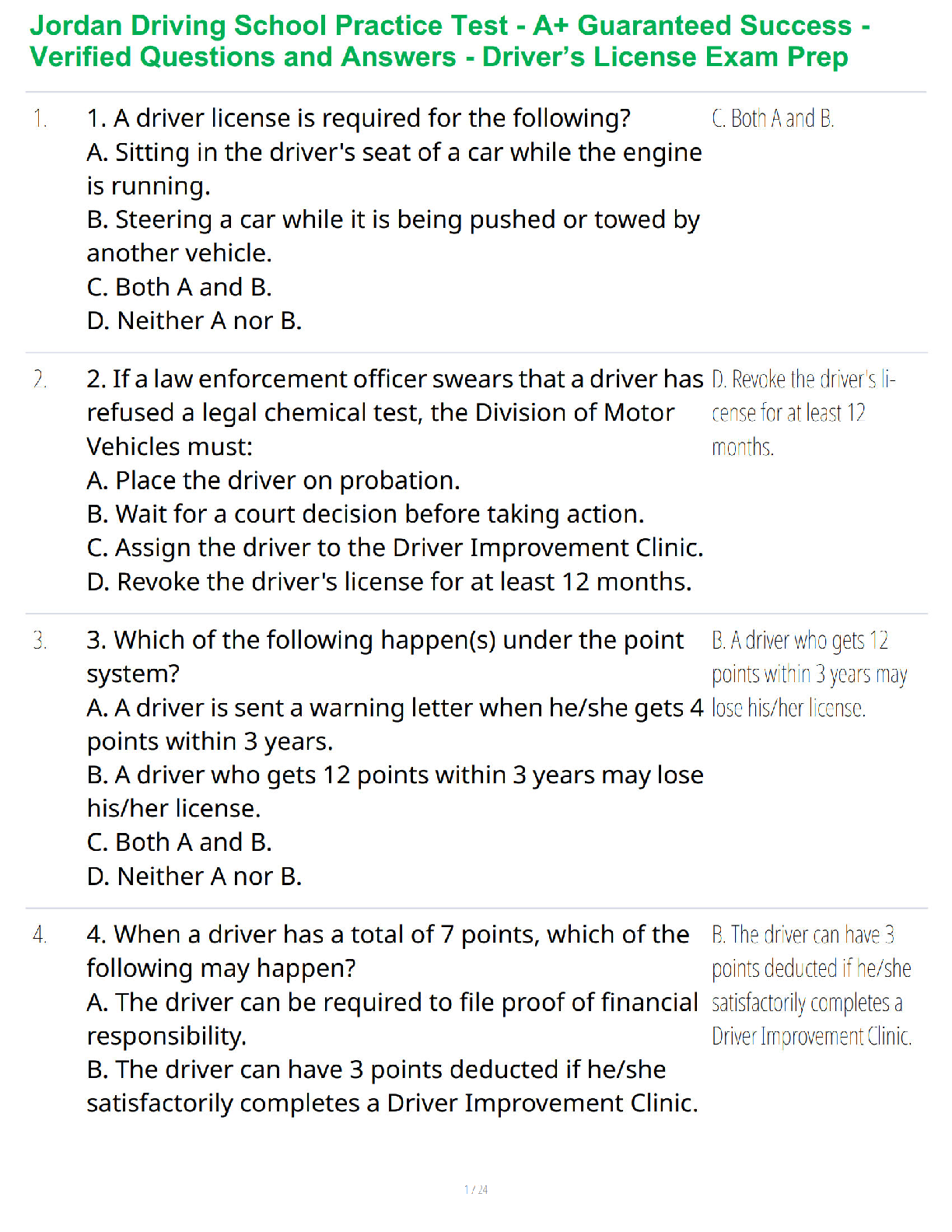
Chap 1. -Positive feedback: Strengthen or reinforce a change in one of the body’s controlled
conditions. Control center provides a physiological response that adds to or reinforces the initial
change in the controlled
...
Chap 1. -Positive feedback: Strengthen or reinforce a change in one of the body’s controlled
conditions. Control center provides a physiological response that adds to or reinforces the initial
change in the controlled condition—continues till interrupted by some mechanism.
Frontal plane: Divides the body into anterior (front) and posterior (back) portions.
Chap 2. -Water molecule bond: One atom of oxygen shares electrons with two atoms of
hydrogen. Polar covalent bond, sharing of electrons is unequal=nucleus of one atom attracts the
shared electrons more strongly than the nucleus of the other atom. Oxygen atom attracts the
electrons more strongly than the hydrogen atoms=oxygen atom has greater electronegativity.
*Inorganic compounds=structurally simple, water/salts/acids/bases, cannot be used by cells to
perform biological functions, carbon dioxide
Organic compounds – always contain carbon(carbon not easily dissolved in water, useful for
body structures), covalent bonds, large molecules. Carbs/lipids/proteins/ATP/nucleic acids.
They carry out complex functions within the body.
*Covalent bonds=can form between atoms of the same element, can be polar or non-polar,
forms when electrons are shared, and most common chemical bond in the body
*Atomic number of an element=proton # in nucleus
Mass number – protons plus neutrons
Isotopes – atoms that have the same number of protons but different numbers of neutrons
(different mass #) most are stable. If an atom is an isotope, its chemical properties remain the
same, this only changes if the electron number is different, as electrons determine chemical
properties of that element.
Atomic mass – amount of all naturally occurring isotopes.
Non-polar bond – a covalent bond where atoms share electrons equally
Polar bond – a covalent bond where atoms do not share electrons equally. One atom has more
electronegativity. (H20 is a polar covalent bond. O attracts electrons more)
Hydrogen bond – partially positive hydrogen attracts partially negative atoms. (usually O or N)
They result from attraction of positive and negative charges. They are weak. They help link
other molecules together.
Enzymes - Properties of enzymes: protein molecules that speed up chemical reactions in
organisms. They are catalysts, they decrease the amount of energy required for a reaction to
occur. Consist of two parts: (1) protein portion (apoenzyme), (2) non protein portion (cofactor).
Three properties: (1) Enzymes are highly specific (enzyme matched to particular substrate), (2)
Enzymes are very efficient, (3) Enzymes are subject to a variety of cellular controls. End in
‘ase’. ATPases split ATP. Proteases break down proteins.
*Fibrous proteins=form muscle fiber, tendons, connective tissue, and bone. (Actin, collagen,
dystrophin, elastin, keratin, myosin, tropomyosin, tubulin, and fibrin). Insoluble in water
*Globular proteins=more water soluble than other classes of proteins and they may have
several functions including transporting, catalyzing, and regulating. Hemoglobin, lipoproteins,
albumin, enzymes, antibodies.
Chap 3. -Facilitated diffusion: when solutes are too polar or highly charged to move through
the lipid bilayer by simple diffusion they can cross the plasma membrane by passive process(no
cell energy) or facilitated diffusion(cell energy). Integral membrane protein assists a specific
substance across the membrane via channel or carrier. (Active processes uses atp – active
transport, transport in vesicles)
-Lipid bilayer: Basic structural framework of the plasma membrane, two back to back layers
made up of three types of lipid molecules—phospholipids, cholesterol, and glycolipids. Lipids
are amphipathic molecules=both polar and non polar parts. Polar part = head = hydrophilic =
water loving. Non-polar = tails = hydrophobic = water fearing. Cytosol on inside, extracellular
fluid on outside. Highly permeable to nonpolar molecules such as oxygen, carbon dioxide, and
steroids, moderately permeable to small uncharged polar molecules like water/urea, and
impermeable to ions and large, uncharged polar molecules like glucose. Uncharged on the
inside of cell
Tonicity – a measure of a solutions ability to change the volume of cells by altering the cells
water content. Hypertonic fluid used for cerebral edema. Sports drinks used to rehydrate are
hypotonic.
Transport in vesicles – active process, three types, receptor mediated endocytosis,
phagocytosis, and bulk phase.
Receptor mediated Endocytosis diagram – steps: binding, vesicle formation, uncoating,
fusion with endosome, recycling of receptors to plasma membrane, degradation in lysosomes.
Transcytosis – movement of substances into a cell, through it, and out of the cell. Occur most
often in endothelial cells (line blood vessels), for materials to move from blood into interstitial
fluid.
-Cell diffusion diagram – simple diffusion, channel-mediated facilitated diffusion, carriermediated facilitated diffusion.
-Membrane proteins: Integral or peripheral—form ion channels or pores/carriers/receptors/are
enzymes/linkers /cell-identity markers.
-Integral proteins: extend into the lipid bilayer and are firmly embedded in it. Most are
transmembrane proteins and span the entire lipid bilayer and protrude into both the cytosol and
extracellular fluid. Amphipathic and are tightly attached to one side of the bilayer by covalent
bonding to fatty acids. Consist of glycoproteins, proteins with carbohydrate groups attached to
the ends that protrude into the extracellular fluid.
-Peripheral proteins: not as firmly embedded in the membrane. Attached to the polar heads of
membrane lipids or to integral proteins at the inner or outer surface of the membrane. Support
plasma membrane, anchor integral proteins, move materials within cells.
- Three main parts of the human cell: plasma membrane, cytoplasm(cytosol/organelles), and
nucleus.
- Cytoplasm: consists of organelles, lysosomes, cytoskeleton (centrosomes that help hold the
cell together), and cytosol.
-Nucleus: stores cells DNA. contains ribosomes, chromatin, nucleolus, and DNA. Has its own
cytoplasm called the nucleoplasm.
-Nucleolus: makes mRNA where they get sent to the nuclear envelope where they are fully
assembled into ribosomes. Formed by proteins, RNA, and DNA.
-Nucleoli – produce ribosomes
*Messenger RNA: directs protein synthesis.
[Show More]



.png)












.png)
.png)